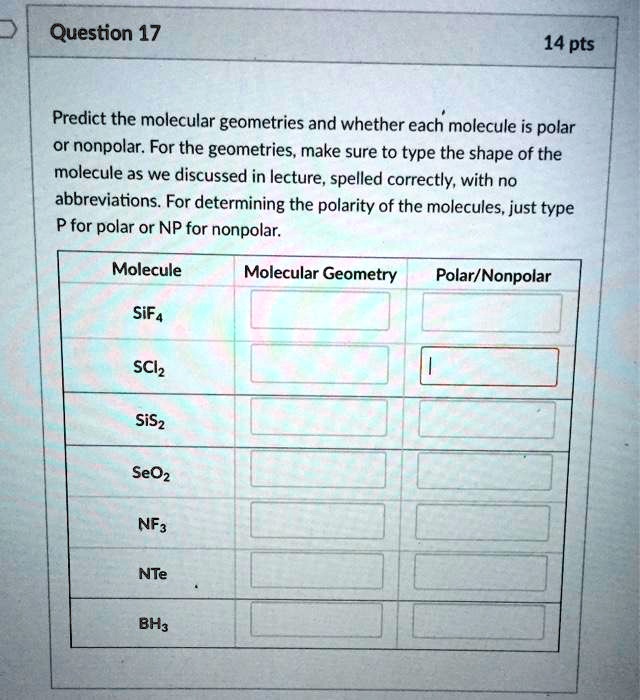
Is NH4+ Polar or Nonpolar? - Polarity of Ammonium ion in 2023 | Molecular shapes, Molecular geometry, Chemistry experiments
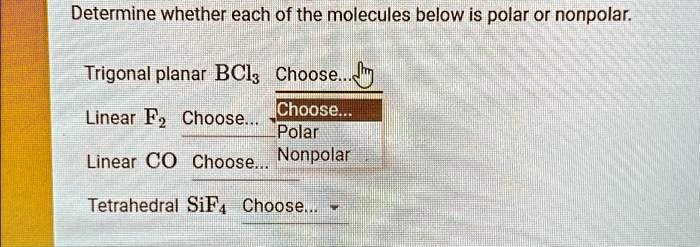
SOLVED: Determine whether each of the molecules below is polar or nonpolar. 1. Trigonal planar BCl3: Choose... 2. Linear F2: Choose... 3. Linear CO: Choose... 4. Tetrahedral SiF4: Choose...

Is the molecule SiF4 polar or nonpolar? If it is polar, identify the atom closest to the negative side. | Homework.Study.com

Is SiF4 polar or non-polar? - Polarity of SiF4 in 2023 | Polar, Molecular geometry, Molecular shapes