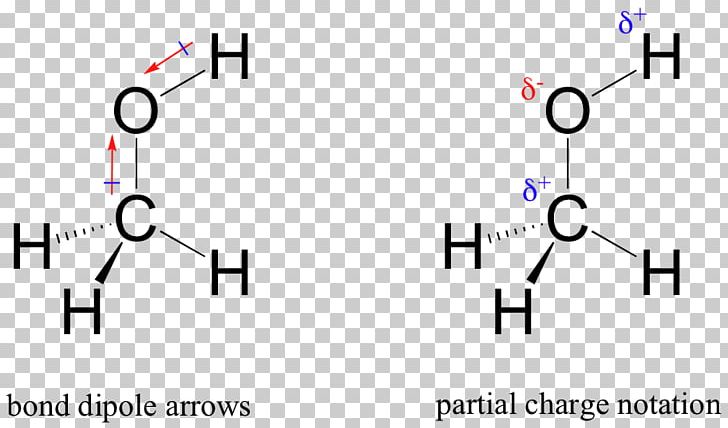
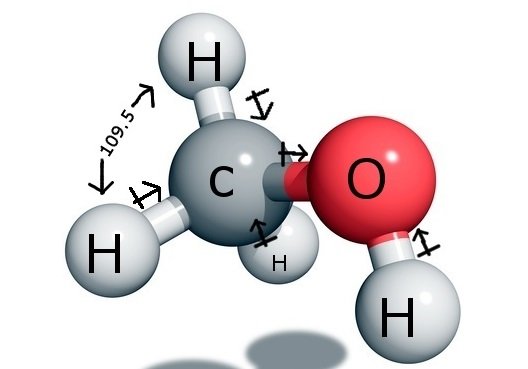
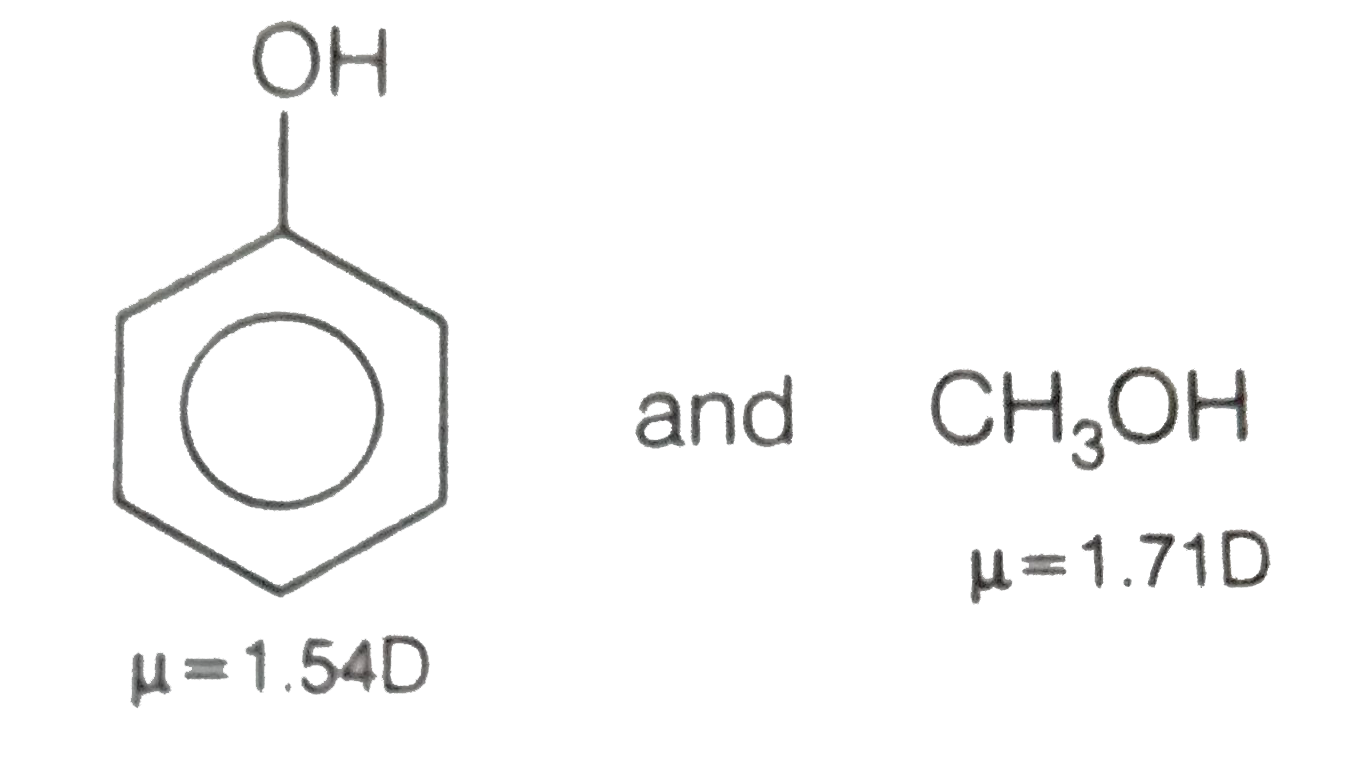
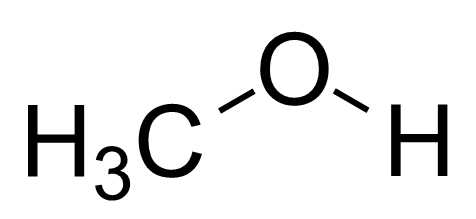
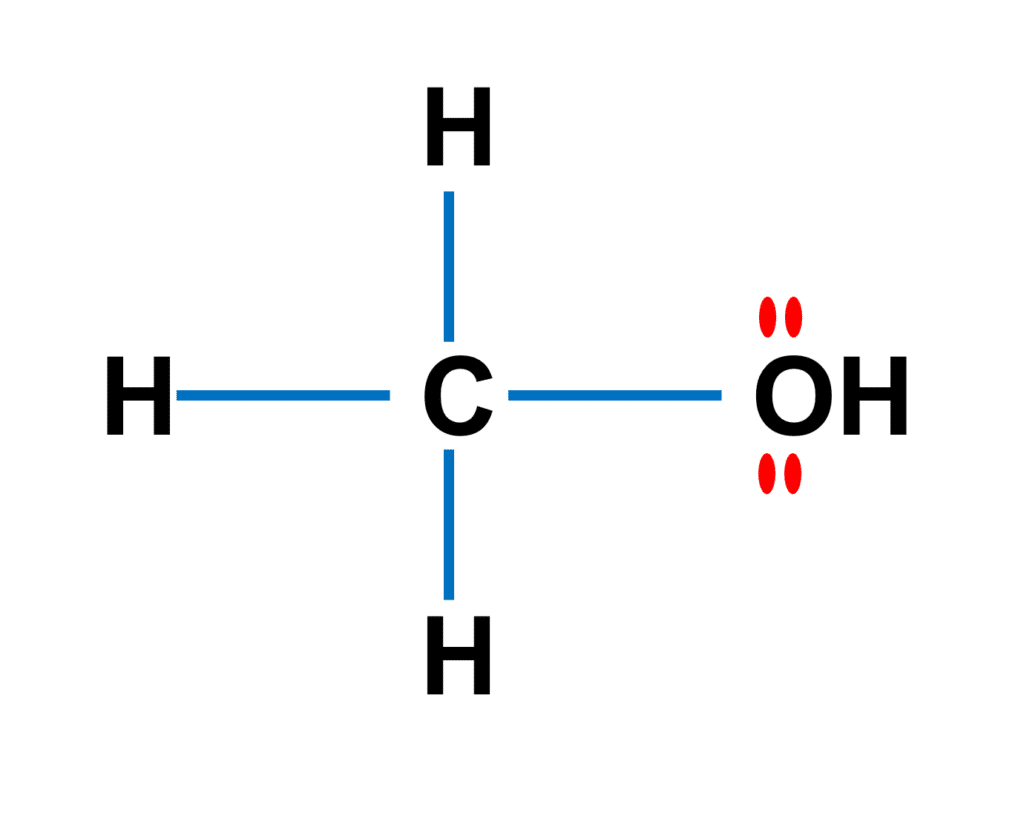
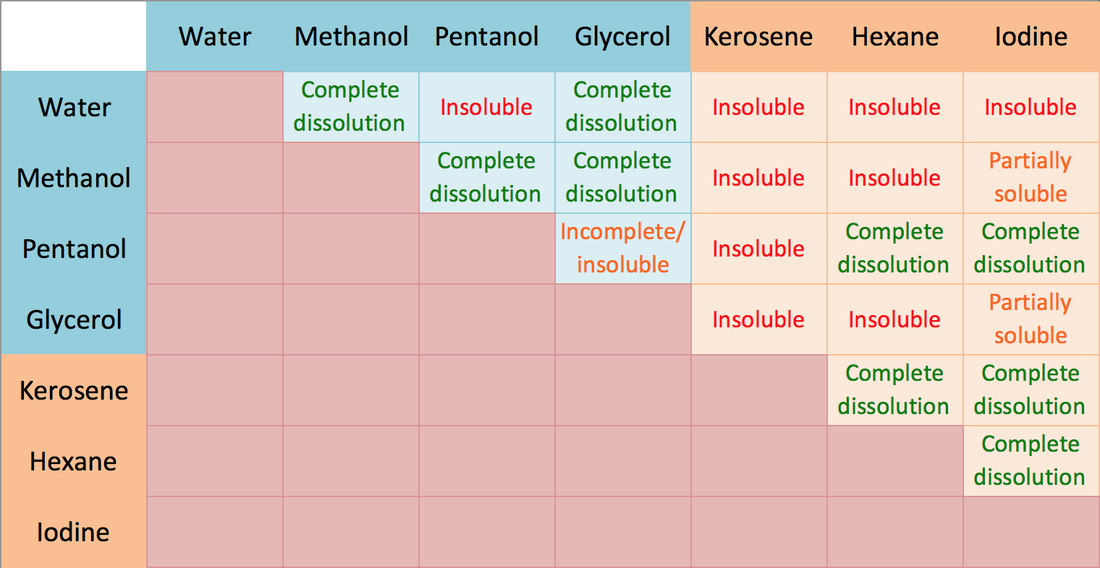
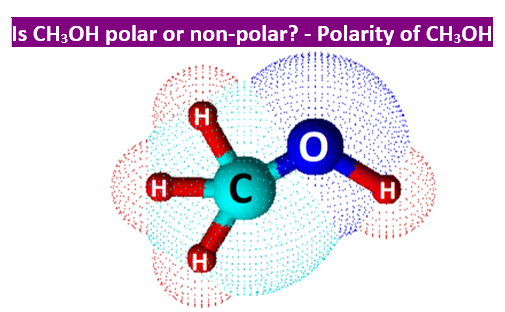
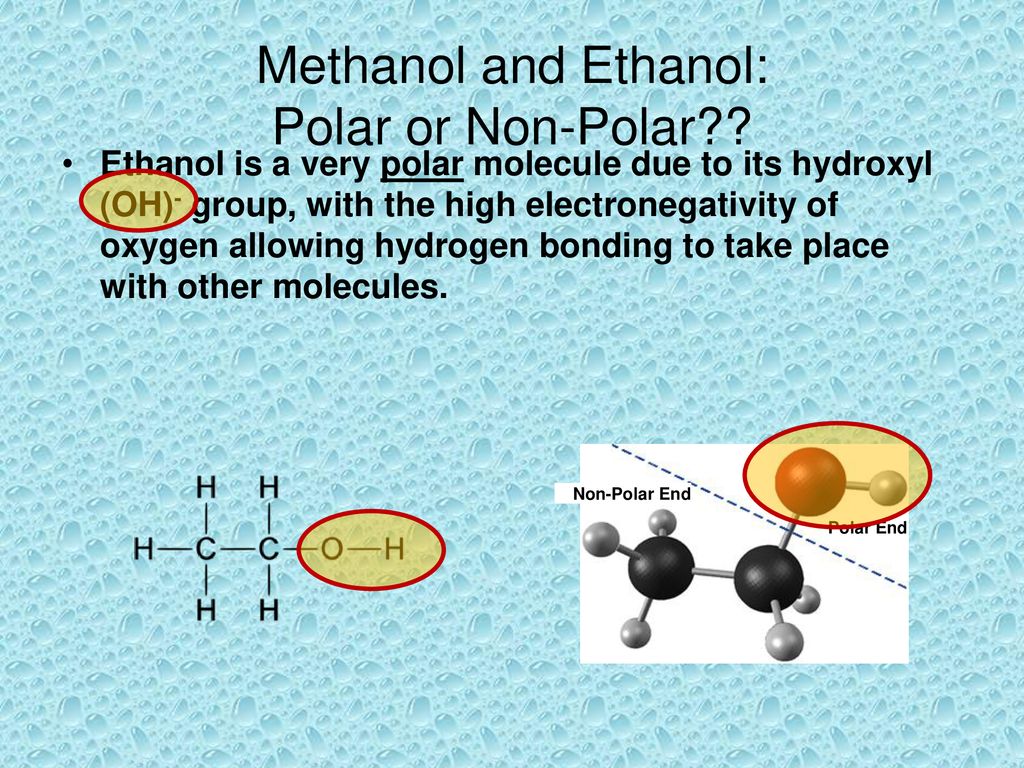
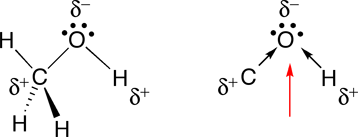SOLVED: Why is methanol (CH3OH) less soluble in hexane (C6H14) than pentanol (I(CH3CH2CH2CH2OH)? Pentanol is a bigger molecule; therefore, hexane can break its bonds more easily. Hexane can form hydrogen bonds with

Chemical polarity Methanol Covalent bond Lewis structure Molecule, induced, angle, white png | PNGEgg