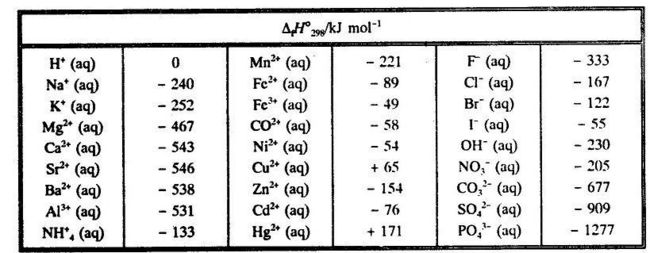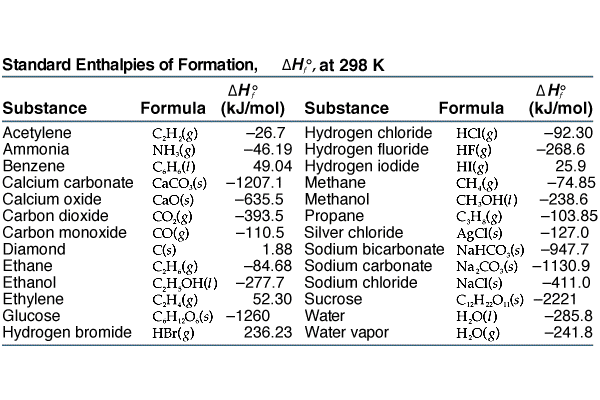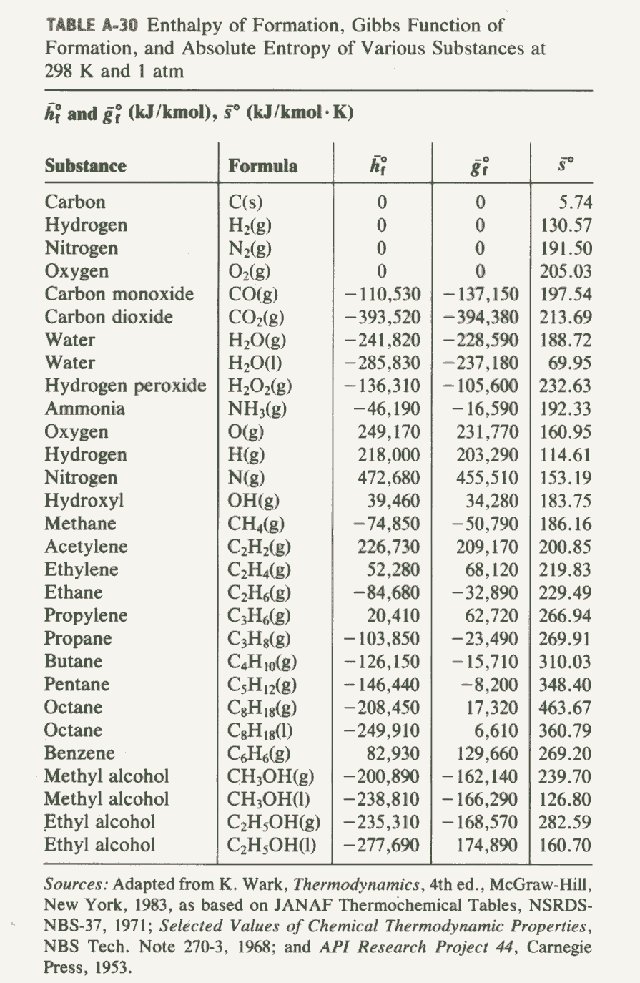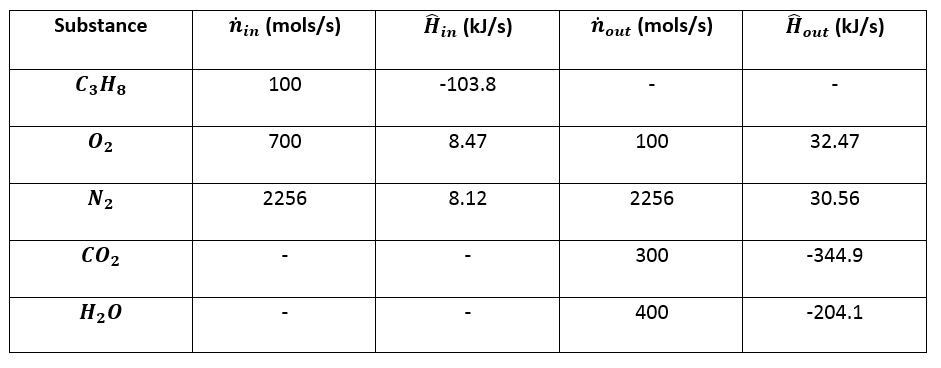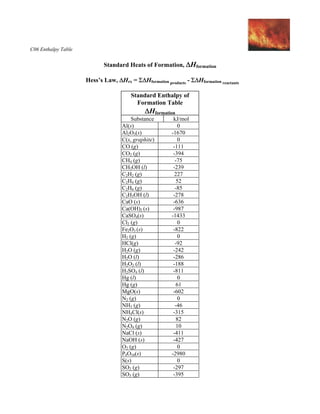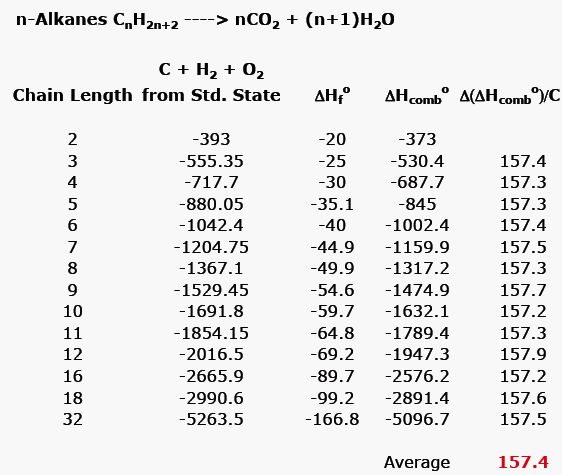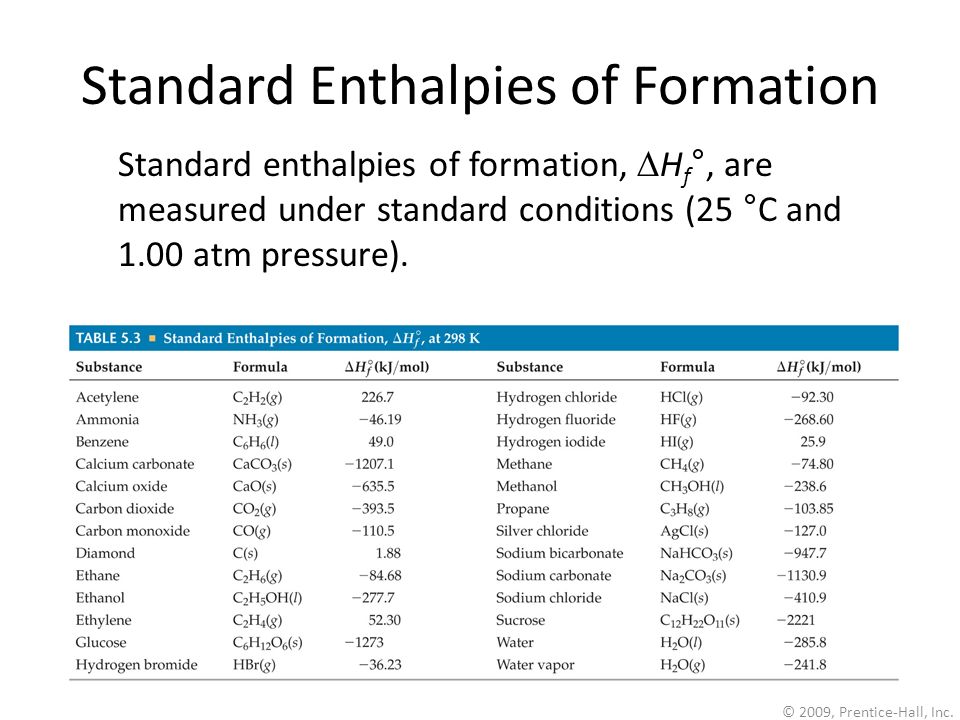
2009, Prentice-Hall, Inc. Enthalpies of Formation An enthalpy of formation, H f, is defined as the enthalpy change for the reaction in which a compound. - ppt download
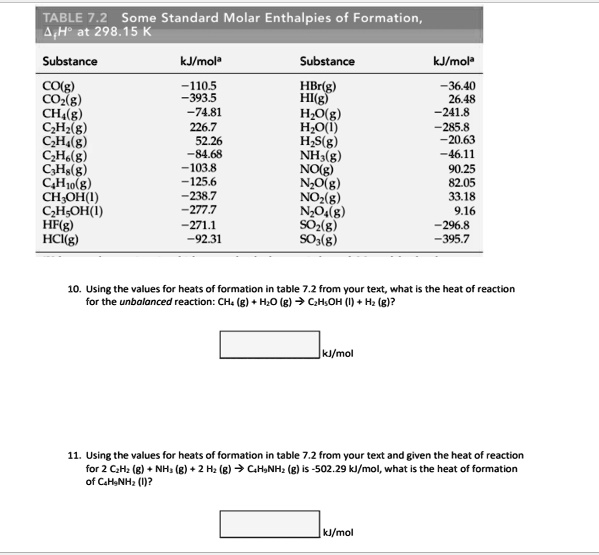
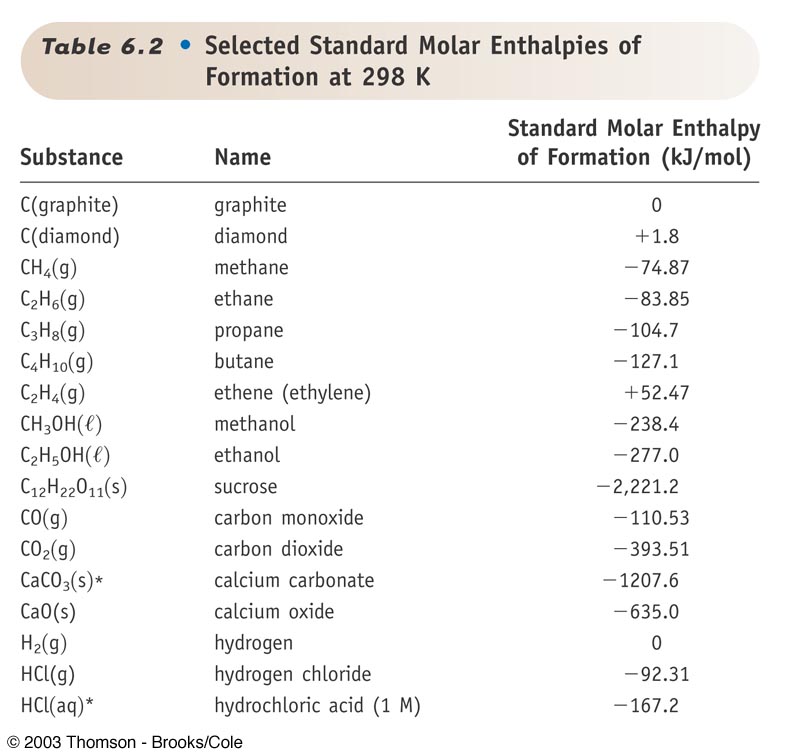
SOLVED: TABLE 7.2 Some Standard Molar Enthalpies of Formation; Heat at 298.15 K Substance kJ/mol Substance kJ/mol CO(g) CO2(g) CH4(g) C2H6(g) C3H8(g) C4H10(g) C6H6(g) C6H12(g) C6H14(g) C6H6OH(l) C2H5OH(l) -105 -393.5 -74.81 226.7

Table 6 from Structure and heats of formation of iodine fluorides and the respective closed-shell ions from CCSD(T) electronic structure calculations and reliable prediction of the steric activity of the free-valence electron
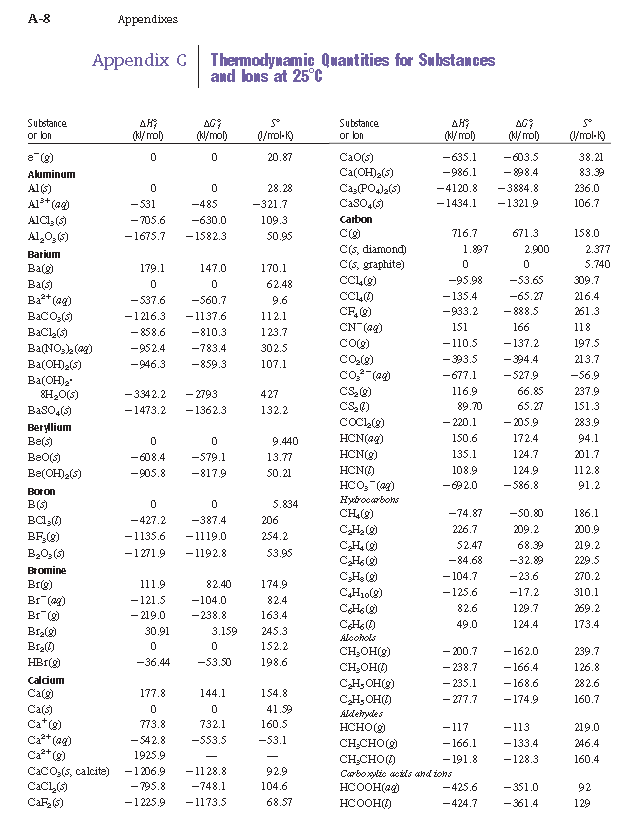
![Appendix D. Heats of Formation and Combustion - Basic Principles and Calculations in Chemical Engineering [Book] Appendix D. Heats of Formation and Combustion - Basic Principles and Calculations in Chemical Engineering [Book]](https://www.oreilly.com/api/v2/epubs/9780132885478/files/graphics/appd-tab-d1a.jpg)
Appendix D. Heats of Formation and Combustion - Basic Principles and Calculations in Chemical Engineering [Book]

Solved) - The value for the standard heat of combustion, ?H° combustion, for... (1 Answer) | Transtutors

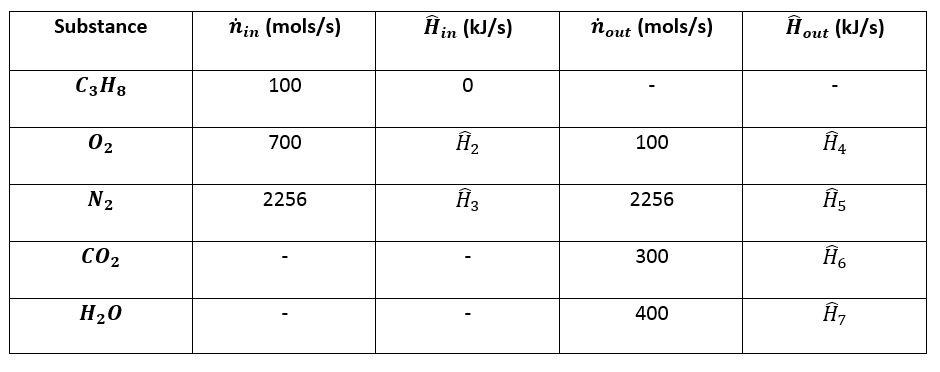
physical chemistry - How do you calculate the heat of a reaction given a table of heat of formation values? - Chemistry Stack Exchange


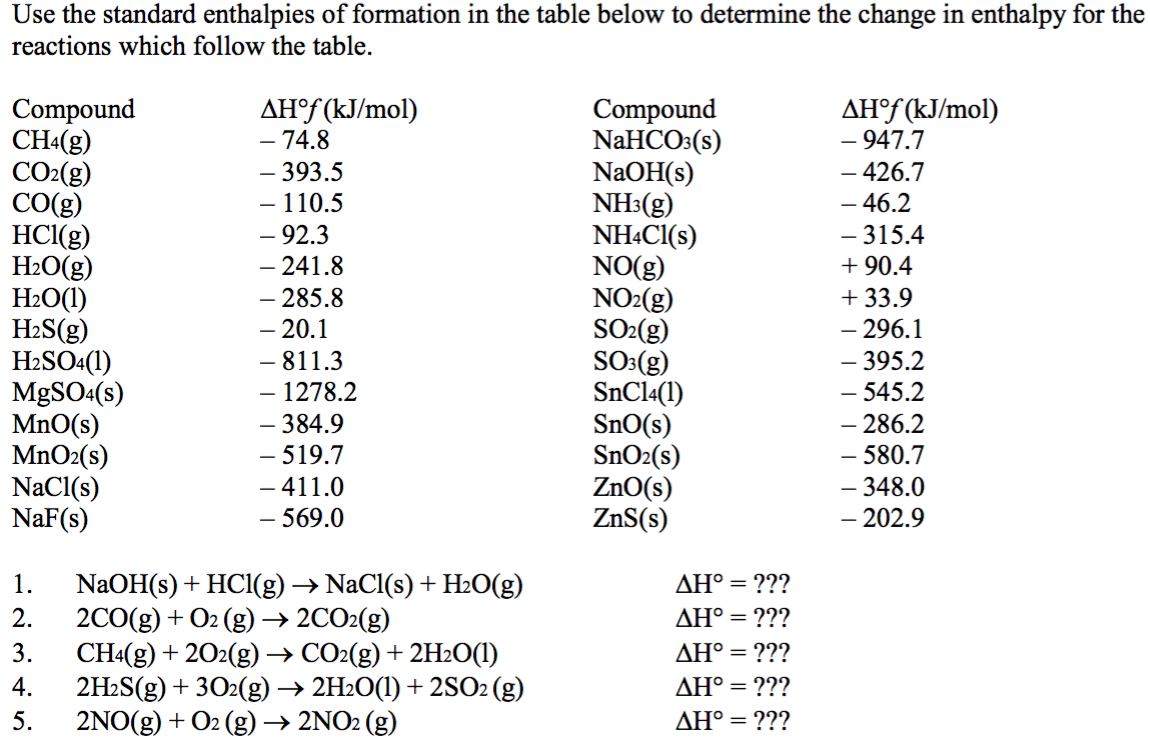



:max_bytes(150000):strip_icc()/GettyImages-154953454-6806780a2f0f4ec99daf580619b5aeef.jpg)