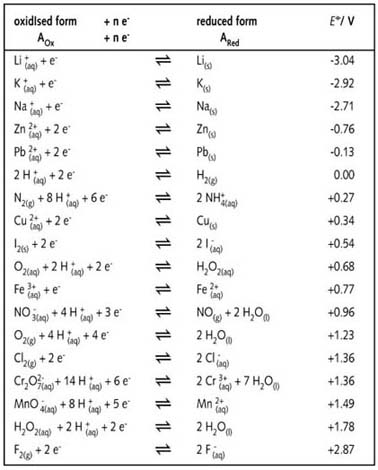
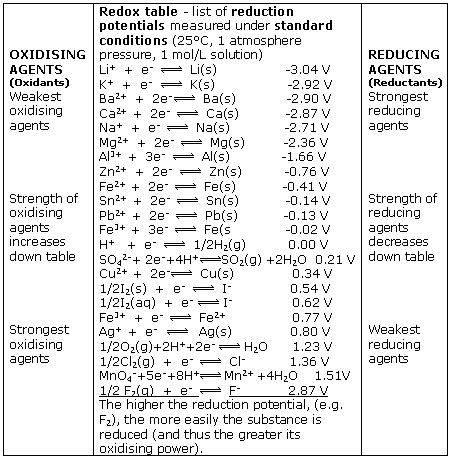
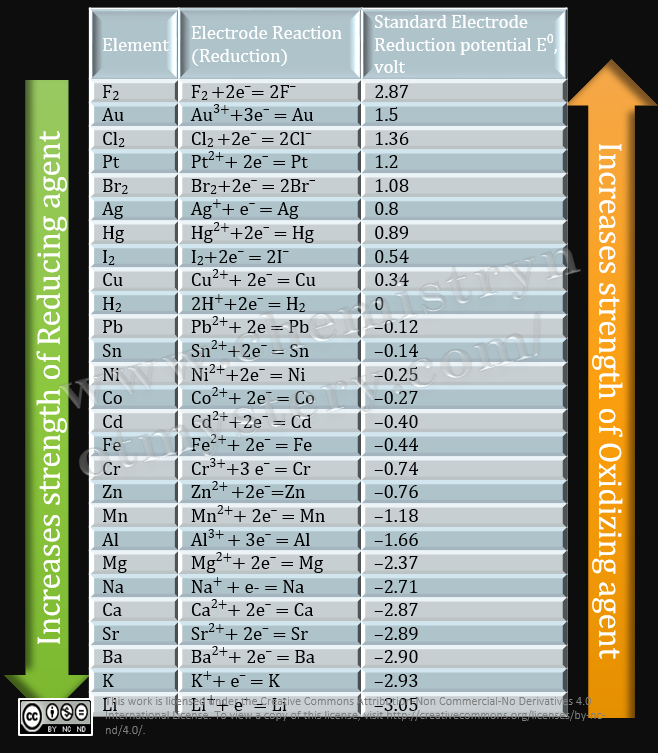
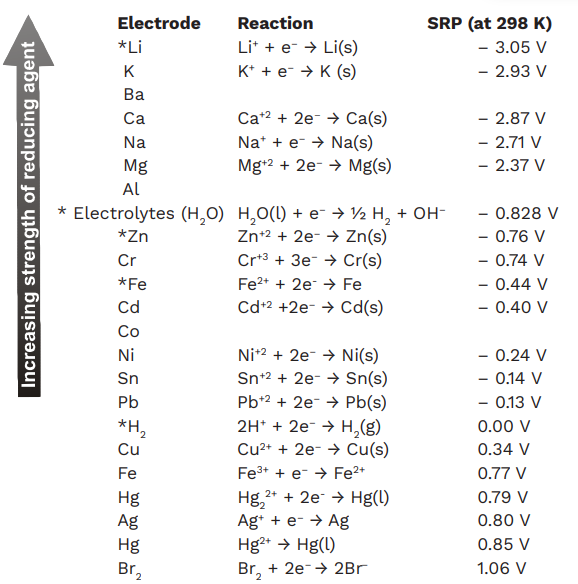
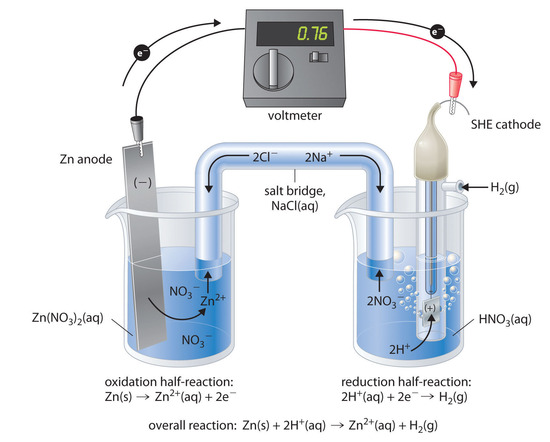
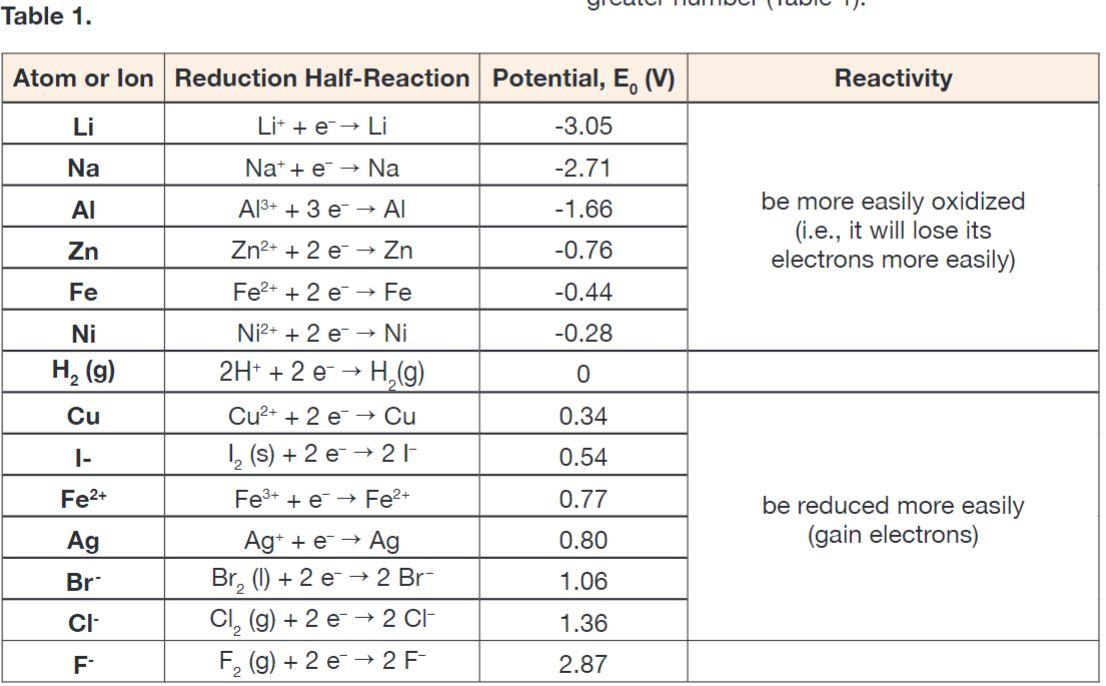
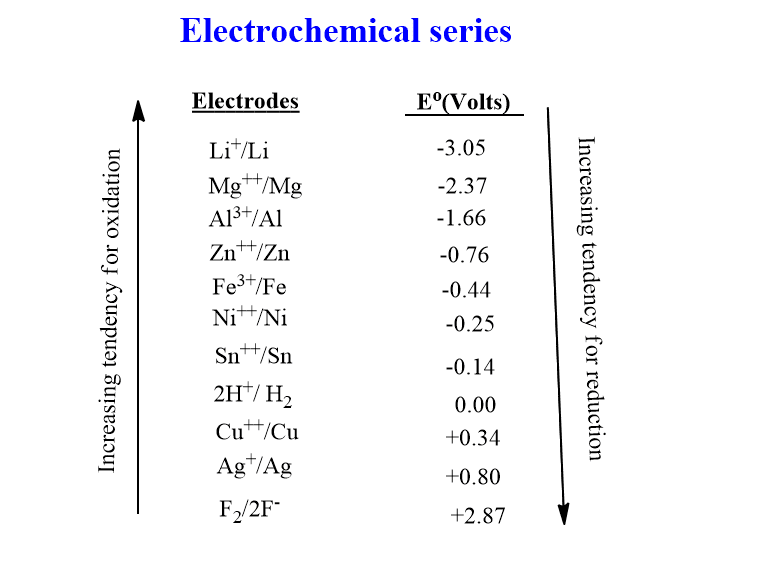
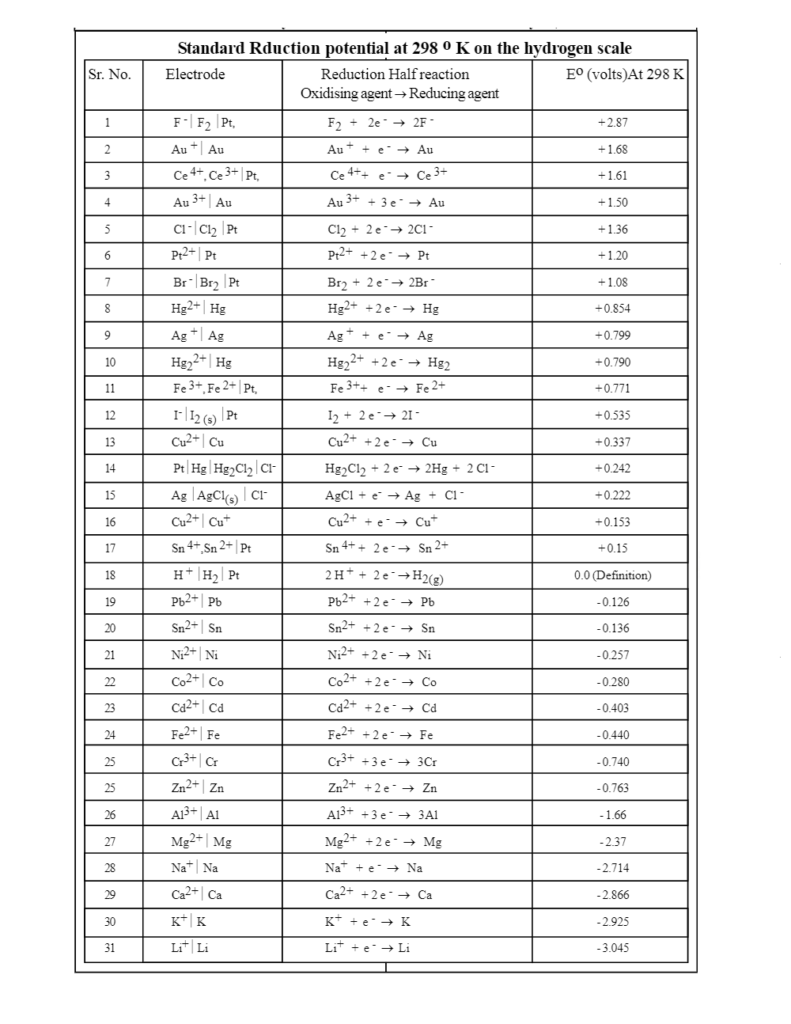
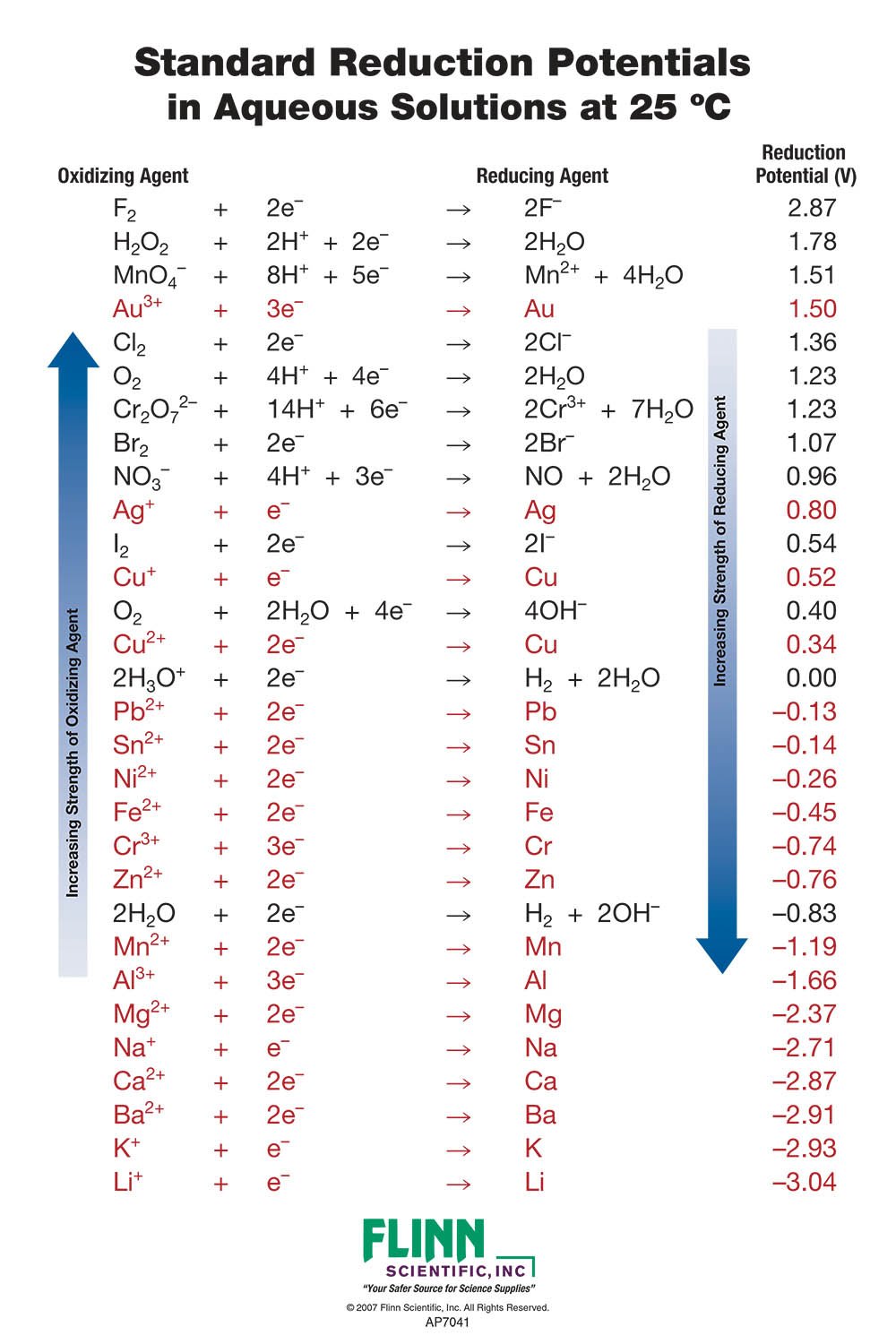
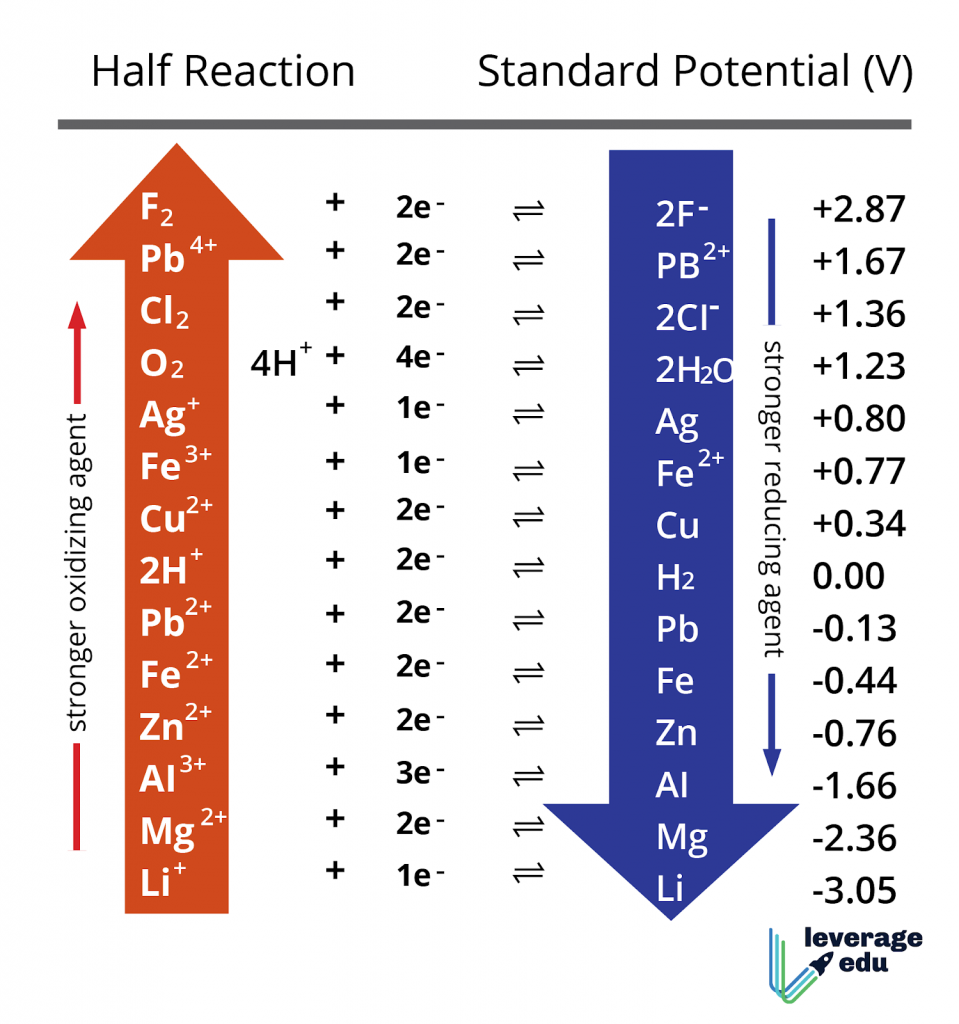
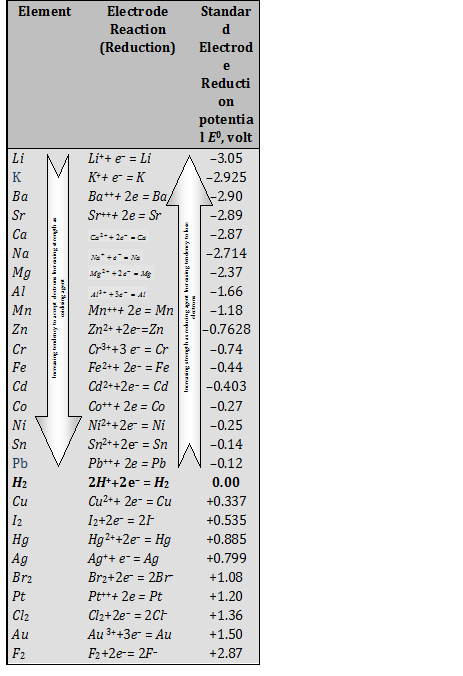
Chemistry - ELECTROCHEMICAL SERIES AND ITS APPLICATION:- A list of elements arranged in order on the basis of their standard reduction potential or oxidation potential is called electrochemical series. EXPLAINATION:- Different elements

Using the standard electrode potentials given in Table 3.1, predict if the reaction between the following is feasible:i Fe 3+ aq and I aq ii Ag + aq and Cu s iii
![PDF] Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K | Semantic Scholar PDF] Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/56964684a624c5af38c7e62256db3faa4c542d88/19-Table2-1.png)





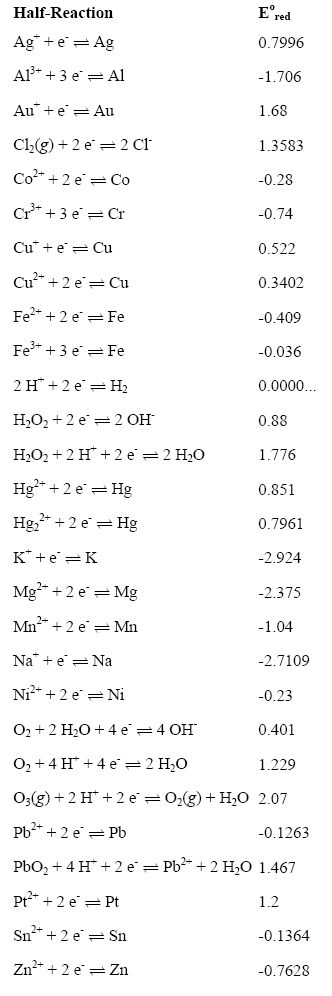
:max_bytes(150000):strip_icc()/Standardreductionpotential-5b551731c9e77c003ec223b3.jpg)
![The variation of the difference of the electrochemical potential [mV]... | Download Table The variation of the difference of the electrochemical potential [mV]... | Download Table](https://www.researchgate.net/publication/279312214/figure/tbl2/AS:667839455326213@1536236742592/The-variation-of-the-difference-of-the-electrochemical-potential-mV-between-the-coat.png)